
Analytical
Science
Regulatory Analytical Science
The Analytical Science teams are a multi-disciplinary group spanning the scientific fields of chromatography, numerous forms of spectroscopy, materials science, and microbiology. The key deliveries of the Analytical Science teams are:
- Qualitative Analysis – determining what substances are present
- Quantitative Analysis – measuring how much of each substance is present
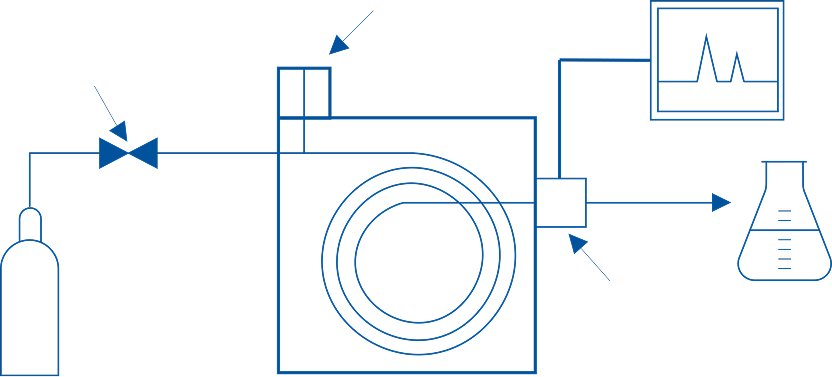
The Analytical Science teams routinely analyze current and future potentially reduced risk products to generate data and interpret results as part of a robust scientific package to satisfy strict Food and Drug Administration (FDA) guidelines for regulatory submissions (Substantial Equivalence (SE), Premarket Tobacco Application (PMTA) and Modified Risk Tobacco Product Application (MRTPA) submissions). This can include information on components and ingredients, as well as stability, chemical, and physical properties.
Our laboratories are ISO 17025 accredited, which is an international standard demonstrating competence of testing and calibration laboratories to operate with consistent accuracy and reliability thus producing valid results for regulatory bodies such as the FDA. This standard requires the Reynolds American organization to meet management and technical requirements, including those related to personnel, facilities, equipment, and standard operating procedures.
Non-Routine Laboratories
The non-routine aspect of Analytical Science provides innovative solutions for investigative research in support of our innovation pipeline to accelerate our Tobacco Harm Reduction goals. Our instrumentation spans a wide array across the fields of chromatography, various spectroscopies, thermal analytical capabilities, microbiological, and bioanalytical technologies.