
Pre-Clinical
Science
OVERVIEW
Our Pre-Clinical assessments follow established regulatory guidelines using standardized testing methods in historical cell and tissue models. This testing is performed to evaluate the potential impact on biological cells and tissues compared to cigarette smoke. The biological testing ranges from assessments performed under Good Laboratory Practices (GLP) guidelines using validated testing methods (e.g., OECD or CORESTA) in support for regulatory submissions to the Food and Drug Administration (FDA) Center for Tobacco Products to exploratory research to further understand our products in support of product development.
Regulatory Toxicological Studies
The following tests are performed to evaluate the potential impact of tobacco and nicotine products on biological cells and tissues.
AMES Assay
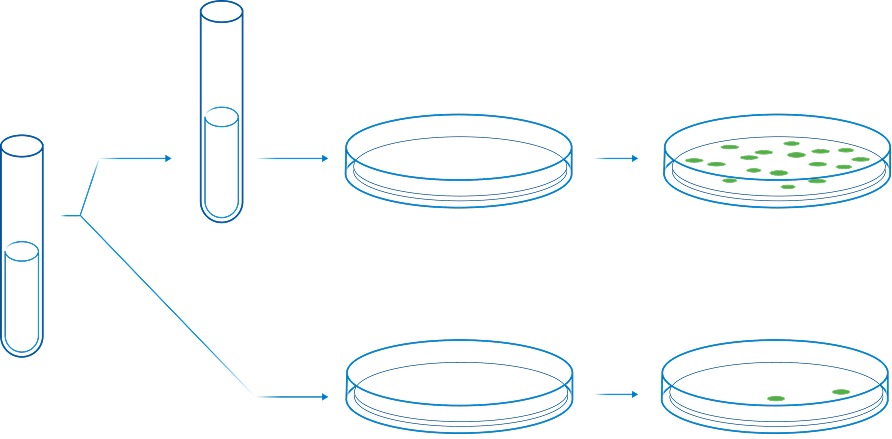
The Ames test is a bacterial assay that uses different strains of bacteria to evaluate the mutagenicity of different chemicals. The Ames Assay was developed in the early 1970s and has been adopted in many different industries worldwide to help identify potentially mutagenic compounds. This Assay has been used extensively to evaluate the mutagenic potential of cigarette smoke.
in vitro Micronucleus (ivMN)
The in vitro micronucleus assay measures the genotoxicity (aneugenic and clastogenic) potential of various chemicals in a cell line. The development of the standardization of this assay began in the early 1970s and was adopted by the OECD in the early 2000s. Adaptation by OECD has led to a wider use of this assay by several industries including the tobacco industry. The in vitro micronucleus assay has been used widely to evaluate the genotoxicity potential of cigarette smoke.
Neutral Red Uptake (NRU)
The neutral red uptake assay evaluates the cytotoxic potential of a chemical by measuring the cell viability following a chemical exposure in vitro. This assay was developed at Rockefeller University in 1984 and was standardized with adaption by the OECD in the early 2000s. The cytotoxicity of cigarette smoke has been evaluated extensively in this assay using a variety of in vitro cell lines.
New Approach Methodologies
The use of New Approach Methodologies (NAMs) aims to provide more human-relevant data to be used to assess risk of exposure. Ongoing research into NAMs include exposure of organotypic cell cultures of the human airway with freshly generated whole smoke or aerosol and measuring a wide variety of different biological and disease-related endpoints.